Which Compound Has the Atom With the Highest Oxidation Number
View solution Oxidation number of iodine varies from. Oxidation number of element in a compound can be positive or negative or may be zero.

Solved Which Compound Has The Atom With The Highest Chegg Com
Sulfur or S has an oxidation number of 4.

. However when bonded with an element with less electronegativity than it it exhibits an oxidation number of -1. By definition the oxidation number of an atom is the charge that atom would have if the compound was composed of ions. View solution Oxidation number of iodine varies from.
In the given compounds the highest oxidation number will be of Magnesium sulfite or MgSO. The most common oxidation numbers are -1 as in HCl and NaCl and 0 as in Cl2. Chemistry questions and answers.
Which compound has the atom with the highest oxidation number. In sodium compounds sodium only forms 1 oxidation number. Which compound has the atom with the highest oxidation number.
Which compound has the atom with the highest oxidation numbera. Sulfur has highest oxidation number for. Oxidation number of each hydrogen atom is.
Cas O NazN MgSO3 AlNO23 - NH4Cl. Chemistry questions and answers. The oxidation number of each atom can be calculated by subtracting the sum of lone pairs and electrons it gains from bonds from the number of valence electrons.
Oxidation state or number of magnesium sulfite can be is calculated as. Among the given compound I shows the highest oxidation state and it is 7. Magnesium or Mg has an oxidation number of 2.
3 AlNO 2 3 NH Cl 4. A Na2CO3 B K2SO4 C FeNO33 D ZnS. The net ionic equation for formation of an aqueous solution of NiI 2 accompanied by evolution of CO2 gas via mixing solid.
MgSO3 CaS NH4Cl Na3N AlNO23. Thus the atoms in O 2 O 3 P 4 S 8 and aluminum metal all have an oxidation number of 0. Which one of the following compounds is insoluble in water.
Identify the element which can have the highest oxidation number. NH4Cl i tbkught ht was MgSO3 but it was marked wrong help i thought. Oxidation number is a property of atoms and not compounds.
MgSO4 What volume of a concentrated solution of sodium hydroxide 600must be diluted to 2000ml to make a 0880 M solution of sodium hydroxide. Oxygen has an oxidation of -2 in most of its compounds. In which of the following compounds does the sulfur atom have the highest ie most positive oxidation number.
Oxygen or O has an oxidation number of-2. The total number of electrons that an atom either gains or loses in order to form a chemical bond with another atom. The hydrogen atom H exhibits an oxidation state of 1.
Now summing up the oxidation states we get. A CaS B Na3N C MgSO3 D AlNO23 E NH4Cl. The oxidation number of an atom is zero in a neutral substance that contains atoms of only one element.
1 1 pts Question 3 Which compound has the atom with the highest oxidation number. Which compound has the atom with the highest oxidation number. Considering this which compound has the atom with the highest oxidation number.
A CaS B Na3N C MgSO3 D AlNO23 E NH4Cl. Yes you are right Sulphur has the highest oxidation number. 33 Which compound has the atom with the highest oxidation number.
Which solution has the same number of moles of NaOH as 500 mL of 0100 M solution of NaOH. In the coordination compound CrNH3en2ClBr2 the coordination number CN and oxidation number ON of the metal atom are respectively asked Aug 30 2019 in Chemistry by Donna general-chemistry. M1V1M2V2 600150 x 200 0880 x 200176600 293.
But some types of atoms such as chlorine form various oxidation numbers like -1 0 1. Which compound has the atom with the highest oxidation number. A 200 mL of 0200 M solution of NaOH.
Which compound has the atom with the highest oxidation number. Therefore Oxidation number of Carbon Oxidation number of Nitrogen Charge on the Cyanide ion x 3 1 x 2 Hydrogen has a 1 oxidation state. CaS Na N 3 MgSO Correct.
Click to see full answer. Aqueous solutions of a compound did not form precipitates with Cl- Br- I- SO42-This compound is. Which compound has the atom with the highest oxidation number.
For monoatomic ions the oxidation number always has the same value as the net charge corresponding to the ion.
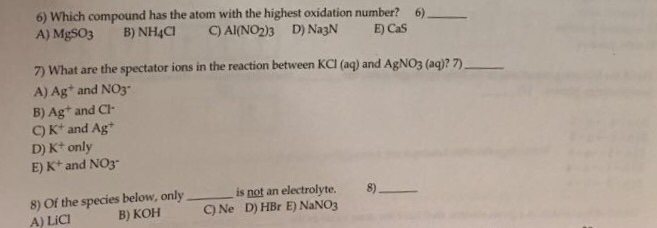
Solved 6 Which Compound Has The Atom With The Highest Chegg Com

Oxidation State Examples Online Chemistry Tutor

Solved 17 Which Compound Has The Atom With The Highest Chegg Com
No comments for "Which Compound Has the Atom With the Highest Oxidation Number"
Post a Comment